JEE Main 2024 8 April Shift 1 Question Paper (Available)- Download Solution PDF with Answer Key
JEE Main 2024 8 April Shift 1 Question Paper with Solutions and Answer Key PDF is available here. NTA conducted JEE Main 2024 8 April Shift 1 exam from 9 AM to 12 PM. The question paper for JEE Main 2024 8 April Shift 1 includes 90 questions equally divided into Physics, Chemistry and Maths. Candidates must attempt 75 questions in a 3-hour time duration. The official JEE Main 2024 question paper pdf for the 8 April Shift 1 exam is available for download using the link below.
Related Links:
- JEE Main 2025 Question Paper pdf with solutions
- JEE Main Previous Years Question Paper with Solution PDF
JEE Main 2024 8 April Shift 1 Question Paper PDF Download
| JEE Main 2024 (Apr 8 Shift 1) Question Paper with Answer Key | Check Solutions |

JEE Main 2024 8 April Shift 1 Questions with Solutions
The value of k ∈ N for which the integral I_n = ∫₀¹ (1 - x^k)^n dx, n ∈ N, satisfies 147I₍₂₀₎ = 148I₍₂₁₎, is:
View Solution
The sum of all solutions of the equation 8^(2x) - 16 · 8^x + 48 = 0 is:
View Solution
Let the circles C₁: (x - a)² + (y - 3)² = r² and C₂: (x - 8)² + (y - 1)² = (r/2)² touch each other externally at the point (6,6). If the point (6,6) divides the line segment joining the centers of the circles C₁ and C₂ internally in the ratio 2:1, then:
(a + b) + 4.(r + r³) equals:
View Solution
The distance of point P from the x-axis, given the conditions:
OP = γ; the angle between OQ and the positive x-axis is θ; and the angle between OP and the positive z-axis is ϕ.
View Solution
The number of critical points of f(x) = (x - 2)^(2/3) (2x + 1) is:
View Solution
The differential equation, whose general solution is y = c₁f(x) + c₂, where c₁ and c₂ are arbitrary constants, is:
View Solution
The number of points of local maxima of f(x) = 4 cos³(x) + 3√3 cos²(x) - 10 in the interval (0, 2π) is:
View Solution
If the variance of the frequency distribution is 160, then the value of c ∈ N is:
View Solution
Let the range of the function f(x) = 1 / (2 + sin(3x) + cos(3x)), where x ∈ R and x ∈ [a, b]. If α and β are respectively the A.M. and G.M. of a and b, then α/β is equal to:
View Solution
Between the following two statements:
Statement-I: Let a = i + 2j − 3k and b = 2i + j − k. Then the vector r satisfying a × r = a × b and a · r = 0 is of magnitude √10.
Statement-II: In a triangle ABC, cos(2A) + cos(2B) + cos(2C) ≥ −3/2.
View Solution
Evaluate the following limit:
lim x → π/2 ∫(π/2)^3 x^3 (sin(2t^(1/3)) + cos(t^(1/3))) dt / (x − π/2)^2
View Solution
The sum of the coefficients of x^(2/3) and x^(-2/5) in the binomial expansion of (x^(2/3) + 1/(2x^(2/5)))^9 is:
View Solution
Let B = [1 3; 1 5] and A be a 2 × 2 matrix such that AB⁻¹ = A⁻¹. If BCB⁻¹ = A and C⁴ + αC² + βI = O, then 2β − α is equal to:
View Solution
If log(e) y = 3 sin^(-1) x, then (1 − x)^2 y′′ − x y′ at x = 1/2 is equal to:
View Solution
The integral ∫(3/4 to 1/4) cos(2 cot^(-1)(√(1 − x) / (1 + x))) dx is equal to:
View Solution
Let a, ar, ar², ... be an infinite G.P. If ∑(n=0 to ∞) ar^n = 57 and ∑(n=0 to ∞) a³r^(3n) = 9747, then a + 18r is equal to:
View Solution
If an unbiased dice is rolled thrice, then the probability of getting a greater number in the i-th roll than the number obtained in the (i−1)-th roll, i = 2, 3, is equal to:
View Solution
The value of the integral ∫(2 to −1) log(e)(x + √(x² + 1)) dx is:
View Solution
Let α, β; α > β, be the roots of the equation x² − √2x − √3 = 0. Let Pn = αⁿ − βⁿ, n ∈ N. Then (11√3 − 10√2)P₁₀ + (11√2 + 10)P₁₁ − 11P₁₂ is equal to:
View Solution
Let a = 2î + αĵ + k̂, b = −î + k̂, c = βĵ − k̂, where α and β are integers and αβ = −6. Let the values of the ordered pair (α, β) for which the area of the parallelogram of diagonals a + b and b + c is √21/2, be (α₁, β₁) and (α₂, β₂). Then α₁ + β₁ − α₂β₂ is equal to:
View Solution
Consider the circle C: x² + y² = 4 and the parabola P: y² = 8x. If the set of all values of α, for which three chords of the circle C on three distinct lines passing through the point (α, 0) are bisected by the parabola P, is the interval (p, q), then (2q − p)² is equal to:
View Solution
Let the set of all values of p, for which f(x) = (p² − 6p + 8)(sin²(2x) − cos²(2x)) + 2(2 − p)x + 7 does not have any critical point, be the interval (a, b). Then 16ab is equal to:
View Solution
For a differentiable function f : R → R, suppose f ′(x) = 3f(x) + α, where α ∈ R, f(0) = 1 and lim x→∞ f(x) = 7. Then, 9f(− log(3)) is equal to:
View Solution
The number of integers between 100 and 1000 having the sum of their digits equal to 14 is:
View Solution
Let A = {(x, y) : 2x + 3y = 23, x, y ∈ N} and B = {x : (x, y) ∈ A}. Then the number of one-one functions from A to B is equal to:
View Solution
Let A, B, and C be three points on the parabola y² = 6x, and let the line segment AB meet the line L through C, parallel to the x-axis, at the point D. Let M and N respectively be the feet of the perpendiculars from A and B on L. Then (AM · BN / CD)² is equal to:
View Solution
The square of the distance of the image of the point (6, 1, 5) in the line x−1/3 = y/2 = z−2/4, from the origin is:
View Solution
If (1/alpha + 1 + 1/(alpha+2) + ... + 1/(alpha+1012)) − (1/2·1 + 1/(4·3) + 1/(6·5) + ... + 1/(2024·2023)) = 1/2024, then alpha is equal to:
View Solution
Let the inverse trigonometric functions take principal values. The number of real solutions of the equation 2sin⁻¹(x) + 3cos⁻¹(x) = 2π/5 is:
View Solution
Consider the matrices A = [2, −5; 3, m], B = [20, m], and X = [x, y]. Let the set of all m, for which the system of equations AX = B has a negative solution (i.e., x < 0 and y < 0), be the interval (a, b). Then 8∫b a |A| dm is equal to:
View Solution
A nucleus at rest disintegrates into two smaller nuclei with their masses in the ratio 2:1. After disintegration, they will move:
View Solution
The following figure represents two biconvex lenses L1 and L2 having focal lengths 10 cm and 15 cm, respectively. The distance between L1 and L2 is:
View Solution
The temperature of a gas is −78°C, and the average translational kinetic energy of its molecules is K. The temperature at which the average translational kinetic energy of the molecules of the same gas becomes 2K is:
View Solution
A hydrogen atom in ground state is given an energy of 10.2 eV. How many spectral lines will be emitted due to the transition of electrons?
View Solution
The magnetic field in a plane electromagnetic wave is:
By = (3.5 × 10-7) sin(1.5 × 103 x + 0.5 × 1011 t) T. The corresponding electric field will be:
View Solution
A square loop of side 15 cm is being moved towards right at a constant speed of 2 cm/s. The front edge enters the 50 cm wide magnetic field at t = 0. The value of induced emf in the loop at t = 10 s will be:
View Solution
Two cars are travelling towards each other at a speed of 20 m/s each. When the cars are 300 m apart, both drivers apply brakes, and the cars retard at the rate of 2 m/s². The distance between them when they come to rest is:
View Solution
The I-V characteristics of an electronic device shown in the figure. The device is:
View Solution
The excess pressure inside a soap bubble is three times the excess pressure inside a second soap bubble. The ratio between the volume of the first and the second bubble is:
View Solution
The de-Broglie wavelength associated with a particle of mass m and energy E is:
λ = h / √(2mE). The dimensional formula for Planck’s constant is:
View Solution
A satellite of 103 kg mass is revolving in a circular orbit of radius 2R. If 104R6 joules of energy is supplied to the satellite, it would revolve in a new circular orbit of radius:
View Solution
The effective resistance between A and B, if the resistance of each resistor is R, will be:
View Solution
Five charges +q, +5q, −2q, +3q, −4q are situated as shown in the figure. The electric flux due to this configuration through the surface S is:
View Solution
A proton and a deuteron (q = +e, m = 2.0u) having the same kinetic energies enter a region of uniform magnetic field B, moving perpendicular to B. The ratio of the radius rd of the deuteron path to the radius rp of the proton path is:
View Solution
UV light of 4.13 eV is incident on a photosensitive metal surface having a work function of 3.13 eV. The maximum kinetic energy of the ejected photoelectrons will be:
View Solution
The energy released in the fusion of 2 kg of hydrogen deep in the sun is EH and the energy released in the fission of 2 kg of 235U is EU. The ratio EH/EU is approximately:
View Solution
A real gas within a closed chamber at 27°C undergoes the cyclic process as shown in the figure. The gas obeys the PV³ = RT equation for the path A to B. The net work done in the complete cycle is (assuming R = 8 J/mol·K):
View Solution
A 1 kg mass is suspended from the ceiling by a rope of length 4 m. A horizontal force F is applied at the midpoint of the rope so that the rope makes an angle of 45° with respect to the vertical axis as shown in the figure. The magnitude of F is:
View Solution
A spherical balloon of radius 1 m is inflated with air at constant temperature. The work done to increase the volume of the balloon by 1 m³ is:
View Solution
Using the work formula for isothermal expansion, the work done is calculated as 4 J.
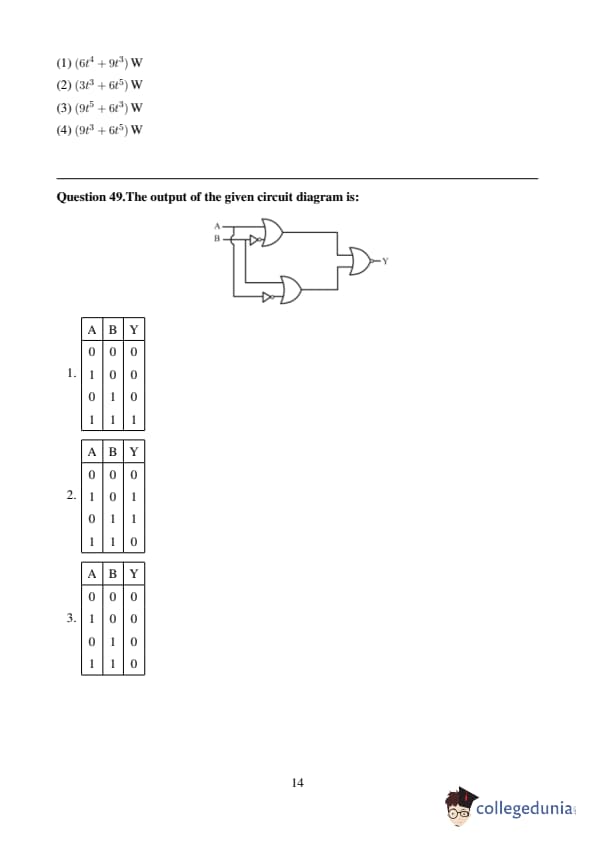
In the truth table of the above circuit, the value of X and Y are:
View Solution
A straight magnetic strip has a magnetic moment of 44 Am². If the strip is bent in a semicircular shape, its magnetic moment will be:
View Solution
A particle of mass 0.5 kg executes simple harmonic motion under a force F = -50x (Nm⁻¹). The time period of oscillation is x/35 seconds. Find the value of x.
View Solution
A capacitor of reactance 4√3 Ω and a resistor of resistance 4 Ω are connected in series with an AC source of peak value 8√2 V. The power dissipation in the circuit is:
View Solution
An electric field E = 2x î N/C exists in space. A cube of side 2 m is placed in the space. The electric flux through the cube is:
View Solution
A circular disc reaches from top to bottom of an inclined plane of length l. When it slips down, it takes t seconds. When it rolls down, it takes (α/2)^1/2 × t seconds. Find α.
View Solution
To determine the resistance (R) of a wire, a circuit is designed. The value of R is:
View Solution
The resultant of two vectors A and B is perpendicular to A and its magnitude is half that of B. The angle between A and B is:
View Solution
Monochromatic light of wavelength 500 nm is used in Young's double-slit experiment. When one slit is covered with a glass plate (refractive index μ = 1.5), the central maximum shifts by 4 fringes. Find the thickness of the glass plate.
View Solution
A force (3x² + 2x - 5) N displaces a body from x = 2 m to x = 4 m. The work done by this force is:
View Solution
At room temperature (27°C), the resistance of a heating element is 50 Ω. If the temperature coefficient of the material is 2.4 × 10⁻⁴ °C⁻¹, find the temperature of the element when its resistance is 62 Ω:
View Solution
The candela is the luminous intensity, in a given direction, of a source that emits monochromatic radiation of frequency A×10¹² hertz and that has a radiant intensity in that direction of 1/B watt per steradian. ’A’ and ’B’ are respectively:
View Solution
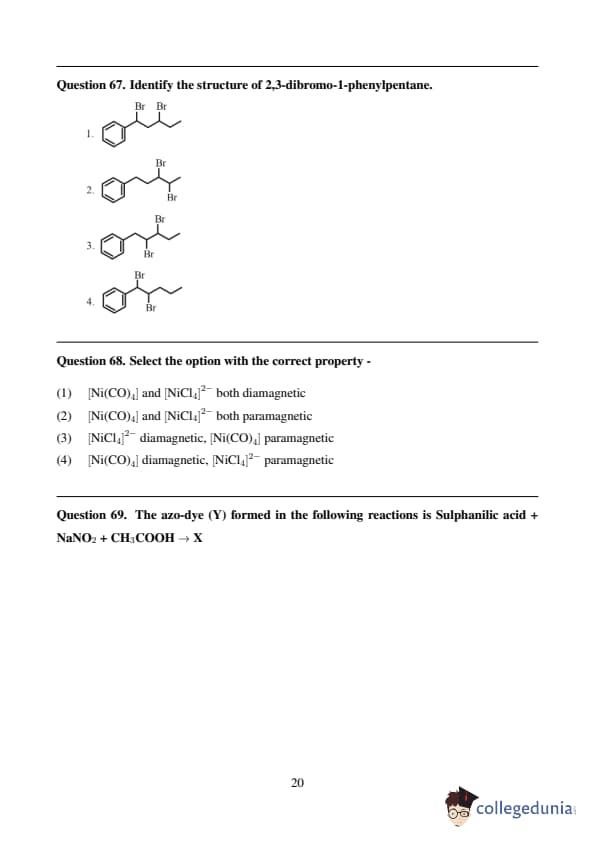
The correct stability order of the following resonance structures of CH₃CH=CHCHO is:
View Solution
The total number of stereoisomers possible for the given structure is:
View Solution
The correct increasing order for bond angles among BF3, PF3, and CF3 is:
View Solution
Match List-I (Test) with List-II (Observation):
List-I (Test) | List-II (Observation)
A. Br2 water test | I. Yellow-orange or orange-red precipitate formed
B. Ceric ammonium nitrate test | II. Reddish orange color disappears
C. Ferric chloride test | III. Red color appears
D. 2,4-DNP test | IV. Blue, green, violet, or red color appears
View Solution
Match List-I (Cell) with List-II (Use/Property/Reaction):
List-I (Cell) | List-II (Use/Property/Reaction)
A. Leclanche cell | I. Converts energy of combustion into electrical energy
B. Ni-Cd cell | II. Does not involve any ion in solution and is used in hearing aids
C. Fuel cell | III. Rechargeable
D. Mercury cell | IV. Reaction at anode: Zn → Zn2+ + 2e-
View Solution
Match List-I (Complex) with List-II (Hybridization):
List-I (Complex) | List-II (Hybridization)
A. K2[Ni(CN)4] | I. sp3
B. [Ni(CO)4] | II. sp3d2
C. [Co(NH3)6]Cl3 | III. dsp2
D. Na3[CoF6] | IV. d2sp3
View Solution
The coordination environment of Ca2+ ion in its complex with EDTA4- is:
View Solution
The incorrect statement about glucose is:
View Solution
The number of oxygen atoms present in the chemical formula of fuming sulfuric acid is:
View Solution
Which of the following compounds can give a positive iodoform test when treated with aqueous KOH solution followed by potassium hypoiodite?
View Solution
For a sparingly soluble salt AB2, the equilibrium concentrations of A2+ ions and B- ions are 1.2 × 10^-4 M and 0.24 × 10^-3 M, respectively. The solubility product of AB2 is:
View Solution
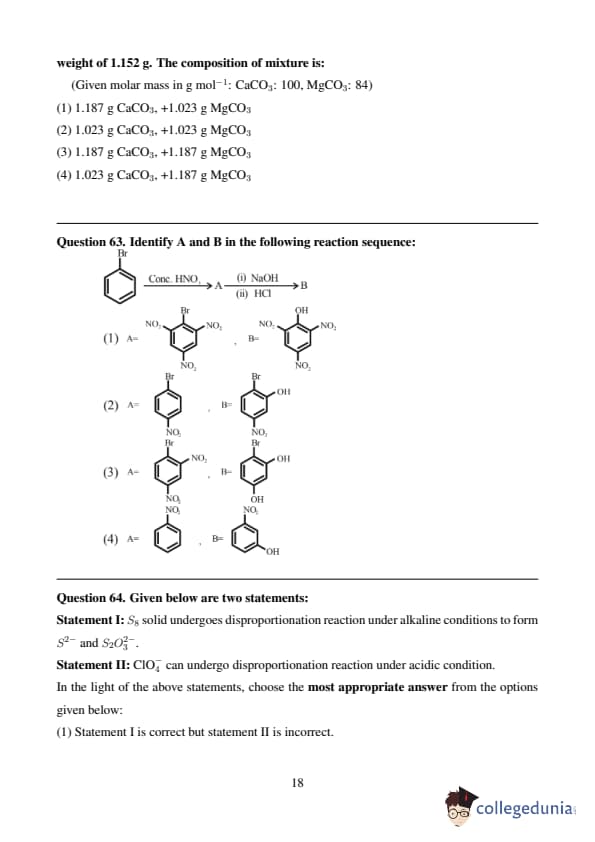
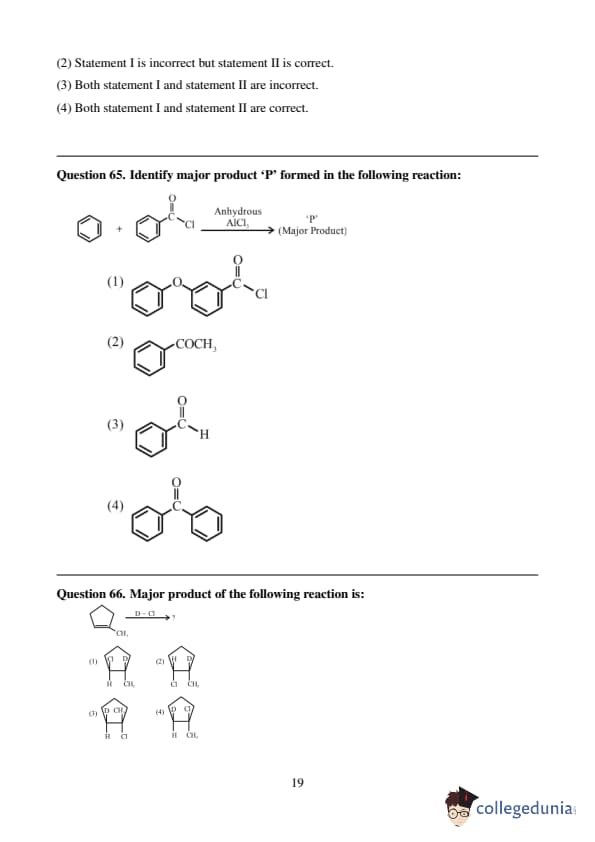
Major product of the following reaction is:
View Solution
Given below are two statements:
Statement I: The higher oxidation states are more stable down the group among transition elements unlike p-block elements.
Statement II: Copper cannot liberate hydrogen from weak acids.
Choose the correct answer from the options given below:
View Solution
The incorrect statement regarding ethyne is:
View Solution
Match List-I with List-II:
List-I (Element) | List-II (Electronic Configuration)
A. N | I. [Ar] 3d10 4s2 4p5
B. S | II. [Ne] 3s2 3p4
C. Br | III. [He] 2s2 2p3
D. Kr | IV. [Ar] 3d10 4s2 4p6
View Solution
Match List-I with List-II:
List-I | List-II
A. Melting point (K) | I. Tl > In > Ga > Al > B
B. Ionic Radius (M+3/pm) | II. B > Tl > Al ≈ Ga > In
C. ∆iH₁ (kJ mol⁻¹) | III. Tl > In > Al > Ga > B
D. Atomic Radius (pm) | IV. B > Al > Tl > In > Ga
View Solution
Which of the following compounds will give a silver mirror with ammoniacal silver nitrate?
View Solution
Which of the following is a correct equation to show change in molar conductivity with respect to concentration for a weak electrolyte, if the symbols carry their usual meaning?
View Solution
The electronic configuration of Einsteinium is: (Given atomic number of Einsteinium = 99)
View Solution
The number of oxygen atoms present in the chemical formula of fuming sulfuric acid is:
View Solution
A transition metal 'M' among Sc, Ti, V, Cr, Mn, and Fe has the highest second ionisation enthalpy. The spin-only magnetic moment value of M⁺ ion is (Nearest integer):
View Solution
The vapor pressure of pure benzene and methyl benzene at 27°C is given as 80 Torr and 24 Torr, respectively. The mole fraction of methyl benzene in the vapor phase, in equilibrium with an equimolar mixture of those two liquids (ideal solution) at the same temperature is ........ ×10⁻² (nearest integer).
View Solution
Consider the following test for a group-IV cation:
M²⁺ + H₂S → A (Black precipitate) + byproduct
A + aqua regia → B + NOCl + S + H₂O
B + KNO₂ + CH₃COOH → C + byproduct
The spin-only magnetic moment value of the metal complex C is ........ BM (Nearest integer).
View Solution
Consider the following first-order gas-phase reaction at constant temperature:
A(g) → 2B(g) + C(g)
If the total pressure of the gases is found to be 200 Torr after 23 sec, and 300 Torr upon the complete decomposition of A after a very long time, then the rate constant of the given reaction is ............. × 10⁻² s⁻¹ (nearest integer).
View Solution
In the given TLC, the distance of spot A and B are 5 cm and 7 cm, from the bottom of the TLC plate, respectively. The Rf value of B is x × 10⁻¹ times more than A. The value of x is:
View Solution
Based on Heisenberg’s uncertainty principle, the uncertainty in the velocity of the electron to be found within an atomic nucleus of diameter 10⁻¹⁵ m is ........ × 10⁹ ms⁻¹ (nearest integer).
View Solution
Number of compounds from the following which cannot undergo Friedel-Crafts reactions is:
Toluene, nitrobenzene, xylene, cumene, aniline, chlorobenzene, m-nitroaniline, m-dinitrobenzene.
View Solution
Total number of electrons present in (π*) molecular orbitals of O₂, O₂⁺, and O₂⁻ is:
View Solution
When ∆Hvap = 30 kJ/mol and ∆Svap = 75 J mol⁻¹K⁻¹, then the temperature of vapor, at one atmosphere, is:
View Solution




































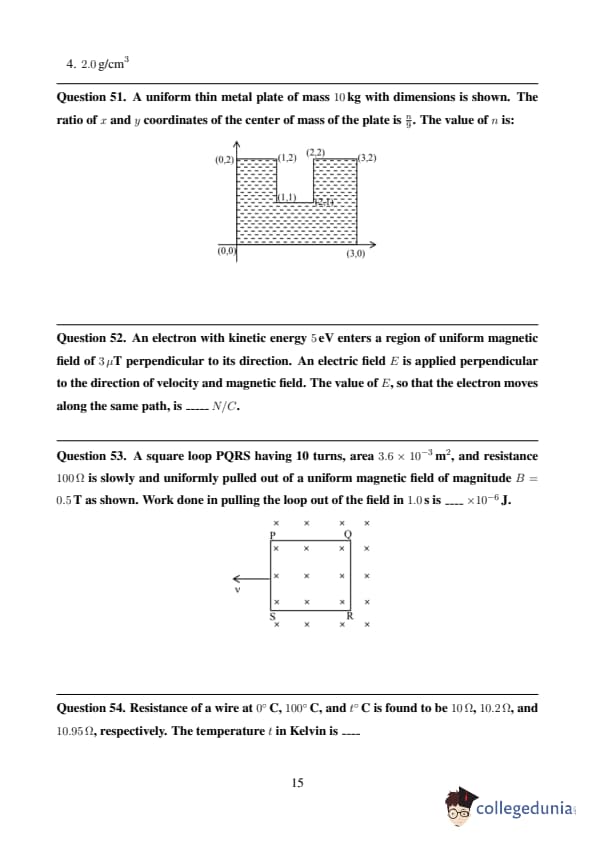





























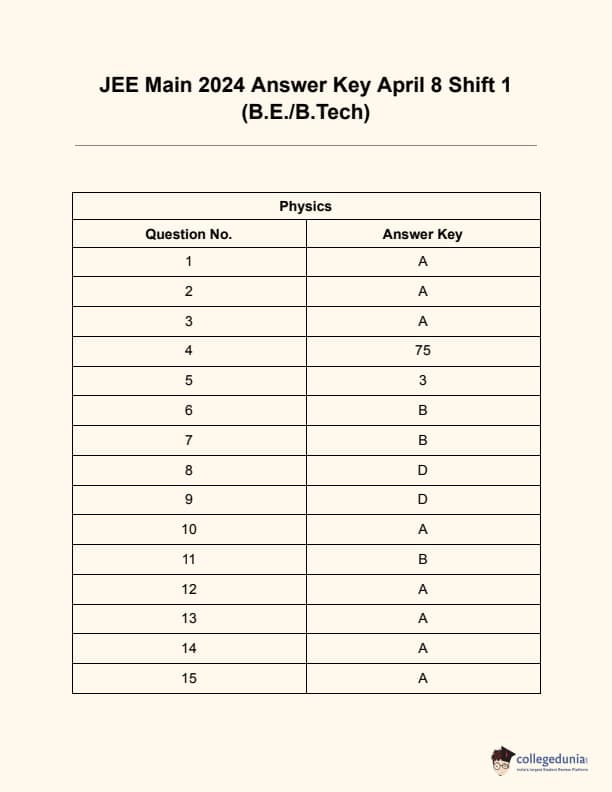
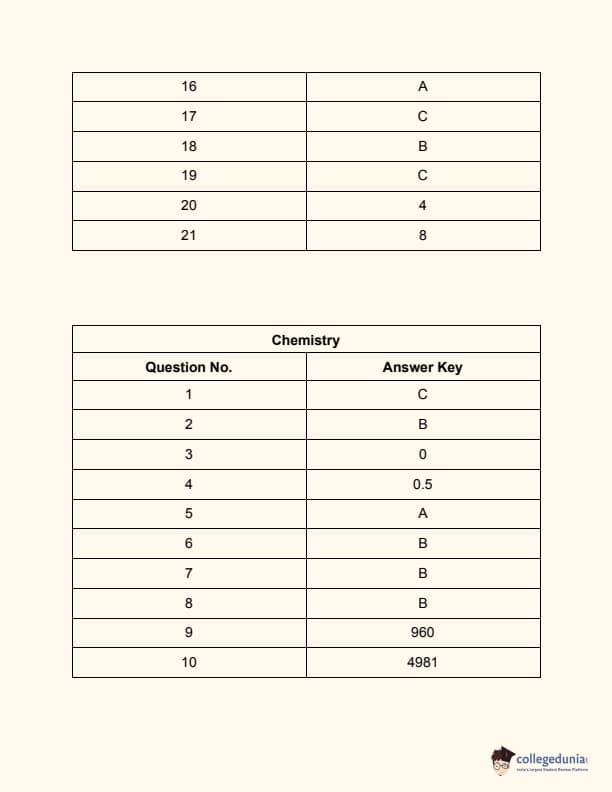
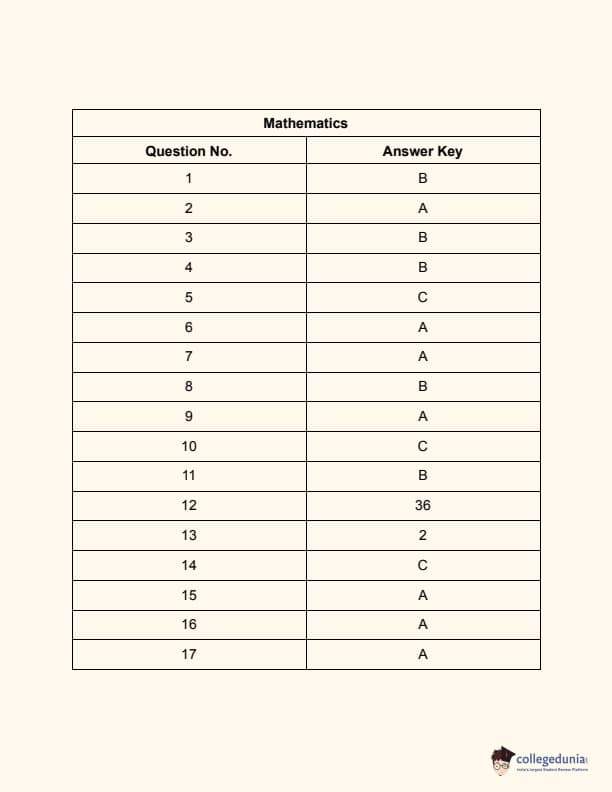
JEE Main 2024 Question Paper Session 2 (April)
Check JEE Main 2024 Question Paper Session 2 for other shifts.
JEE Main 2024 Question Paper Session 1 (January)
Those appearing for JEE Main 2024 can use the links below to practice and keep track of their exam preparation level by attempting the shift-wise JEE Main 2024 question paper provided below.
| Exam Date and Shift | Question Paper PDF |
|---|---|
| JEE Main 27 Jan Shift 1 2024 Question Paper | Check Here |
| JEE Main 27 Jan Shift 2 2024 Question Paper | Check Here |
| JEE Main 29 Jan Shift 1 2024 Question Paper | Check Here |
| JEE Main 29 Jan Shift 2 2024 Question Paper | Check Here |
| JEE Main 30 Jan Shift 1 2024 Question Paper | Check Here |
| JEE Main 30 Jan Shift 2 2024 Question Paper | Check Here |
| JEE Main 31 Jan Shift 1 2024 Question Paper | Check Here |
| JEE Main 31 Jan Shift 2 2024 Question Paper | Check Here |
| JEE Main 1 Feb Shift 1 2024 Question Paper | Check Here |
| JEE Main 1 Feb Shift 2 2024 Question Paper | Check Here |
Also Check:
| JEE Main 2024 Paper Analysis | JEE Main 2024 Answer Key |
| JEE Main 2024 Cutoff | JEE Main 2024 Marks vs Rank |
JEE Main 2024 8 April Shift 1 Question Paper by Coaching Institute
| Coaching Institutes | Question Paper with Solutions PDF |
|---|---|
| Aakash BYJUs | Download PDF |
| Vedantu | Download PDF |
| Reliable Institute | To be updated |
| Resonance | To be updated |
| Sri Chaitanya | To be updated |
| FIIT JEE | To be updated |
JEE Main 2024 8 April Shift 1 Paper Analysis
JEE Main 2024 8 April Shift 1 paper analysis for B.E./ B.Tech is updated here with details on the difficulty level of the exam, topics with the highest weightage in the exam, section-wise difficulty level, etc
JEE Main 2024 Question Paper Pattern
| Feature | Question Paper Pattern |
|---|---|
| Examination Mode | Computer-based Test |
| Exam Language | 13 languages (English, Hindi, Assamese, Bengali, Gujarati, Kannada, Malayalam, Marathi, Odia, Punjabi, Tamil, Telugu, and Urdu) |
| Number of Sections | Three- Physics, Chemistry, Mathematics |
| Exam Duration | 3 hours |
| Sectional Time Limit | None |
| Total Marks | 300 marks |
| Total Number of Questions Asked | 90 Questions |
| Total Number of Questions to be Answered | 75 questions |
| Type of Questions | MCQs and Numerical Answer Type Questions |
| Section-wise Number of Questions | Physics- 20 MCQs and 10 numerical type, Chemistry- 20 MCQs and 10 numerical type, Mathematics- 20 MCQs and 10 numerical type |
| Marking Scheme | +4 for each correct answer |
| Negative Marking | -1 for each incorrect answer |
Read More:
- JEE Main 2024 question paper pattern and marking scheme
- Most important chapters in JEE Mains 2024, Check chapter wise weightage here
Comments